Understanding Pressure Temperature Charts: A Comprehensive Guide
What are Pressure Temperature Charts?
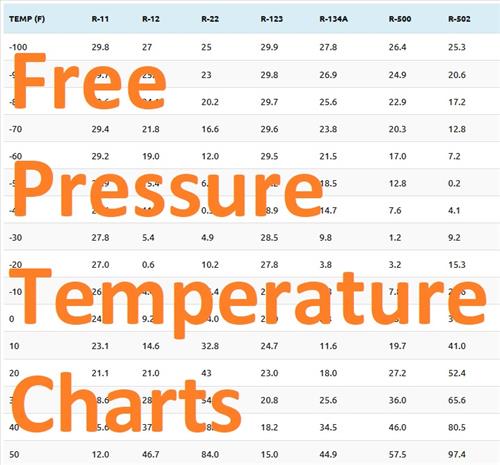
Pressure temperature charts are graphical representations that show the relationship between pressure and temperature of a substance, typically a gas or a liquid. These charts are widely used in various industries, including engineering, chemistry, and physics, to understand the behavior of substances under different conditions. By analyzing pressure temperature charts, professionals can determine the boiling point, condensation point, and other critical properties of a substance, which is essential for designing and operating systems safely and efficiently.
The use of pressure temperature charts is crucial in many applications, such as refrigeration systems, steam power plants, and chemical processing plants. For instance, in refrigeration systems, pressure temperature charts help technicians to determine the optimal operating conditions for the system, ensuring that it runs efficiently and effectively. Similarly, in steam power plants, these charts are used to monitor the pressure and temperature of steam, which is critical for maintaining the plant's safety and performance.
How to Read and Interpret Pressure Temperature Charts
What are Pressure Temperature Charts? Pressure temperature charts are typically plotted with pressure on the vertical axis and temperature on the horizontal axis. The chart shows the vapor-liquid equilibrium curve, which separates the vapor and liquid phases of the substance. The curve indicates the temperature at which the substance changes state from liquid to vapor or vice versa at a given pressure. By analyzing the curve, professionals can determine the critical properties of the substance, such as the critical pressure and critical temperature.
How to Read and Interpret Pressure Temperature Charts To read and interpret pressure temperature charts, one needs to understand the different regions of the chart and how to use them to determine the properties of the substance. The chart is typically divided into three regions: the vapor region, the liquid region, and the two-phase region. By identifying the region in which the substance operates, professionals can determine its state and properties, such as density, specific heat, and viscosity. With this information, they can design and operate systems safely and efficiently, ensuring optimal performance and minimizing the risk of accidents or equipment failure.