Printable Periodic Table Electronegativity: A Comprehensive Guide
What is Electronegativity?
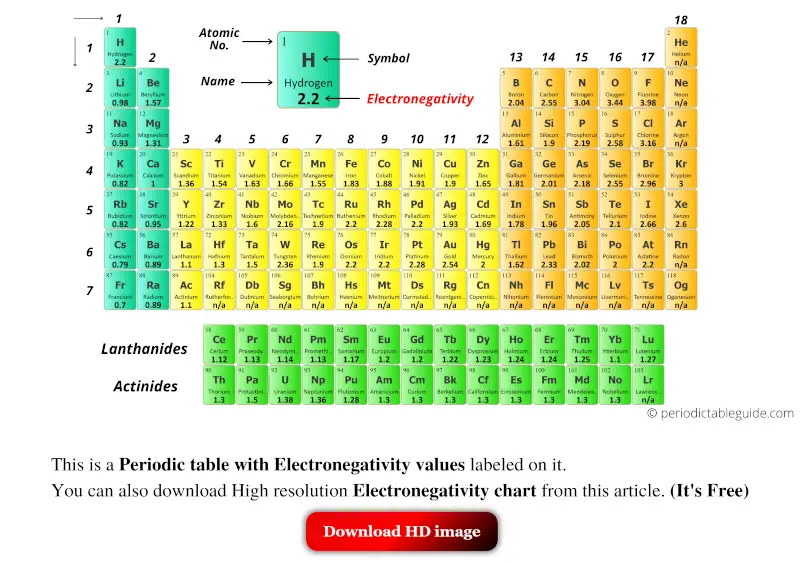
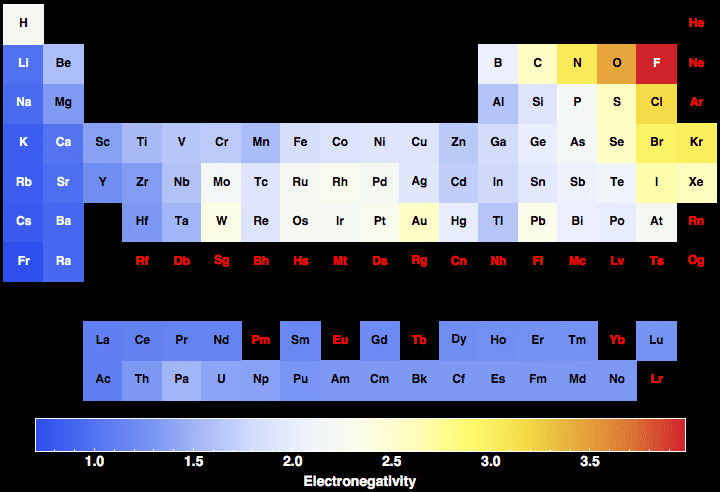
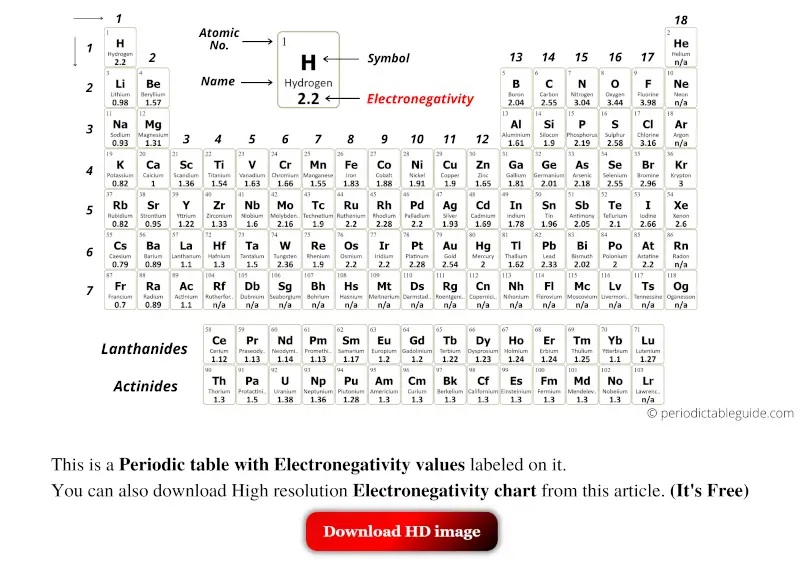
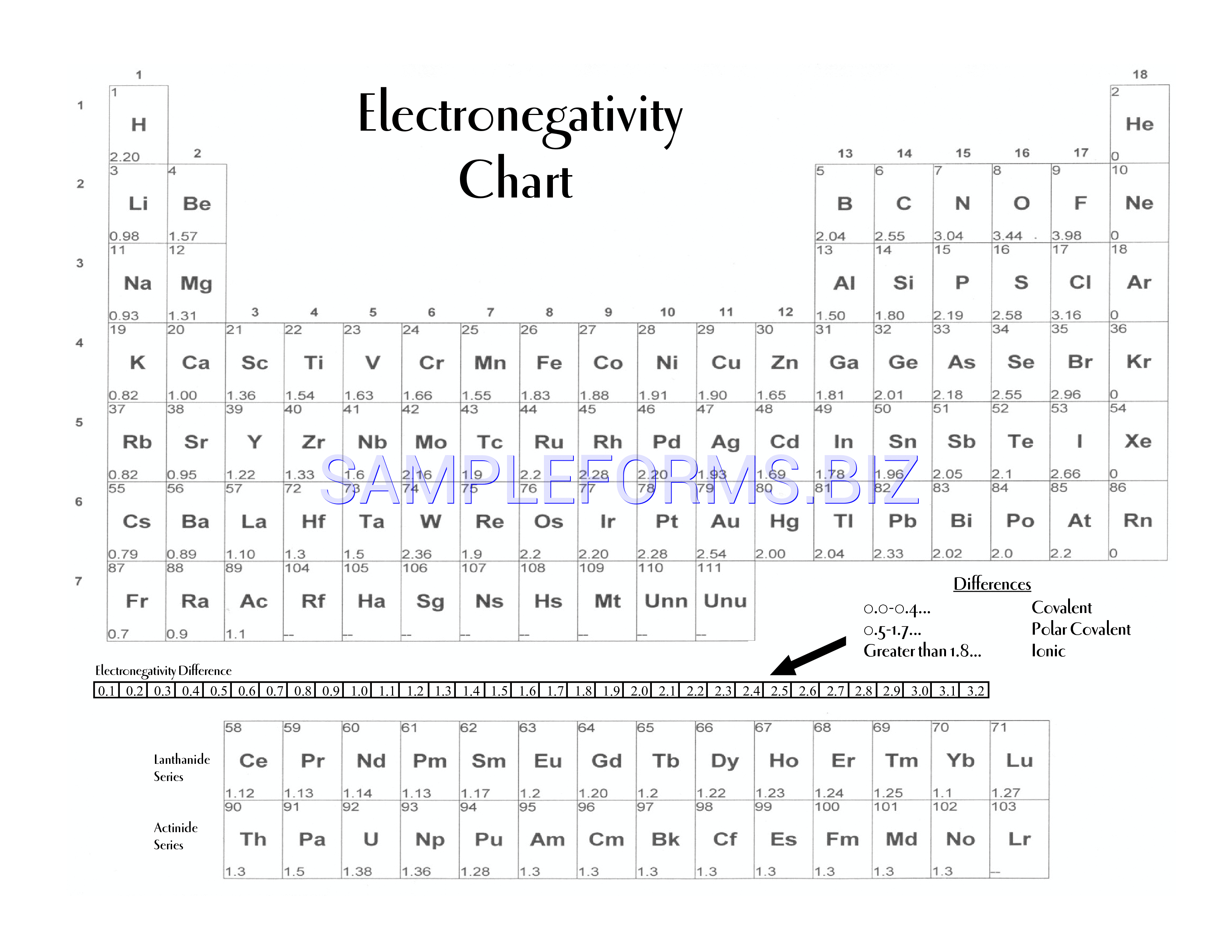
The periodic table is a fundamental tool in chemistry, and one of its most important aspects is electronegativity. Electronegativity is a measure of an atom's ability to attract electrons in a covalent bond. It is a crucial concept in understanding chemical properties and bonding. A printable periodic table with electronegativity values can be a valuable resource for students, teachers, and researchers alike.
The electronegativity of an atom is typically represented by a numerical value, with higher values indicating a greater ability to attract electrons. By using a printable periodic table with electronegativity values, you can easily compare the electronegativity of different elements and understand how they interact with each other. This can be particularly useful in predicting the polarity of molecules and understanding chemical reactions.
Using the Printable Periodic Table
What is Electronegativity? Electronegativity is a measure of an atom's ability to attract electrons in a covalent bond. It is typically represented by a numerical value, with higher values indicating a greater ability to attract electrons. The electronegativity of an atom is influenced by the number of protons in its nucleus and the number of electrons in its outermost energy level. By understanding electronegativity, you can gain insights into the chemical properties and behavior of different elements.
Using the Printable Periodic Table A printable periodic table with electronegativity values can be a valuable tool in the classroom or laboratory. By printing out a copy of the periodic table, you can easily reference the electronegativity values of different elements and use them to predict the polarity of molecules and understand chemical reactions. You can also use the table to identify trends and patterns in electronegativity across different elements and groups, which can help you to better understand the underlying chemistry.