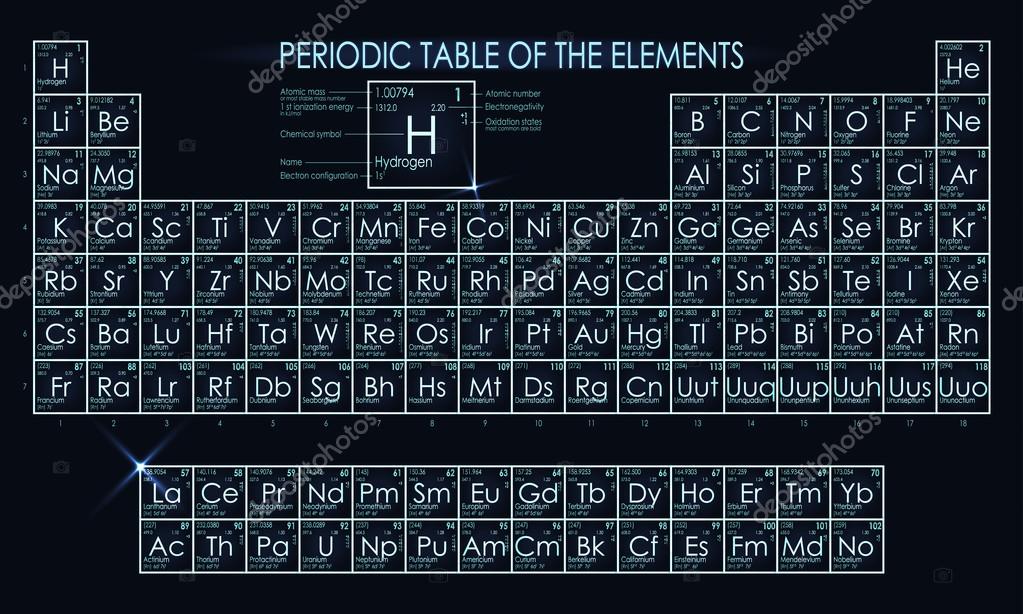
Printable Periodic Table Neon: A Comprehensive Guide
Introduction to Neon
The periodic table is a tabular display of the known chemical elements, and one of the most interesting elements is neon. Neon is a noble gas that is known for its bright, glowing signs and its use in lighting. In this article, we will explore the world of neon and provide a printable periodic table for your reference. Neon is the tenth most abundant element in the universe and is found in the Earth's atmosphere. It is a colorless, odorless, and tasteless gas that is highly unreactive.
Neon has many interesting properties that make it useful for a variety of applications. It has a high ionization energy, which means that it can withstand high temperatures and pressures without breaking down. This makes it ideal for use in neon signs, which are made by electrifying glass tubes filled with neon gas. The printable periodic table can help you understand the properties of neon and its position in the periodic table.
Uses and Applications of Neon
Neon is also used in other applications, such as plasma TVs, neon lasers, and cryogenics. Its high cooling capacity makes it useful for cooling superconducting materials and other applications that require extremely low temperatures. The printable periodic table can help you learn more about the properties and uses of neon, as well as its history and discovery. Neon was discovered in 1898 by Scottish chemist William Ramsay and English chemist Morris Travers, who isolated it from the air.
In conclusion, neon is a fascinating element with many interesting properties and uses. From neon signs to plasma TVs, neon is an essential part of our daily lives. The printable periodic table can help you learn more about neon and its position in the periodic table. Whether you are a student, teacher, or simply interested in learning more about chemistry, a printable periodic table is a valuable resource that can help you understand the world of elements. With its many uses and applications, neon is an element that continues to capture our imagination and inspire new discoveries.